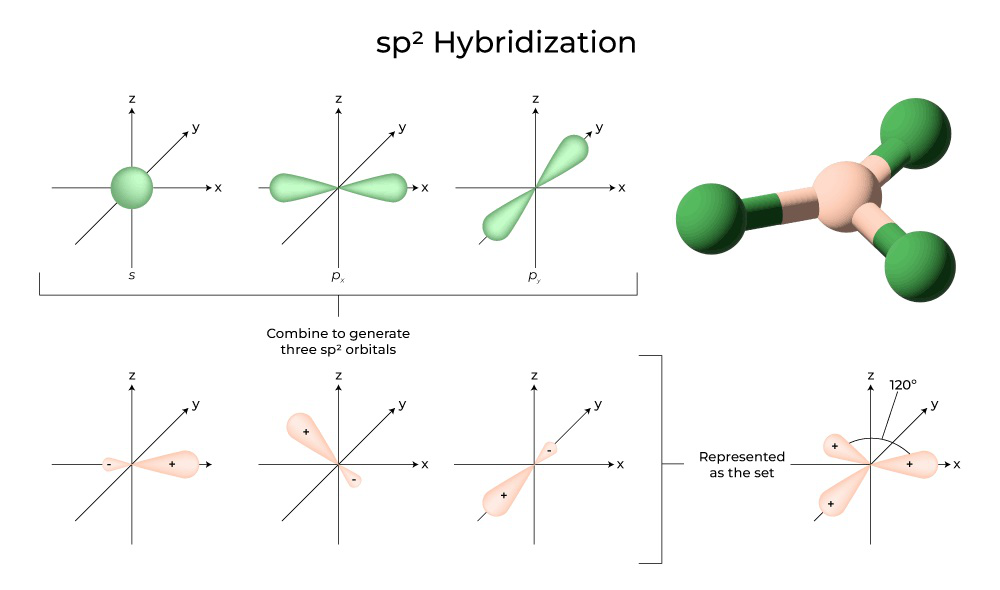
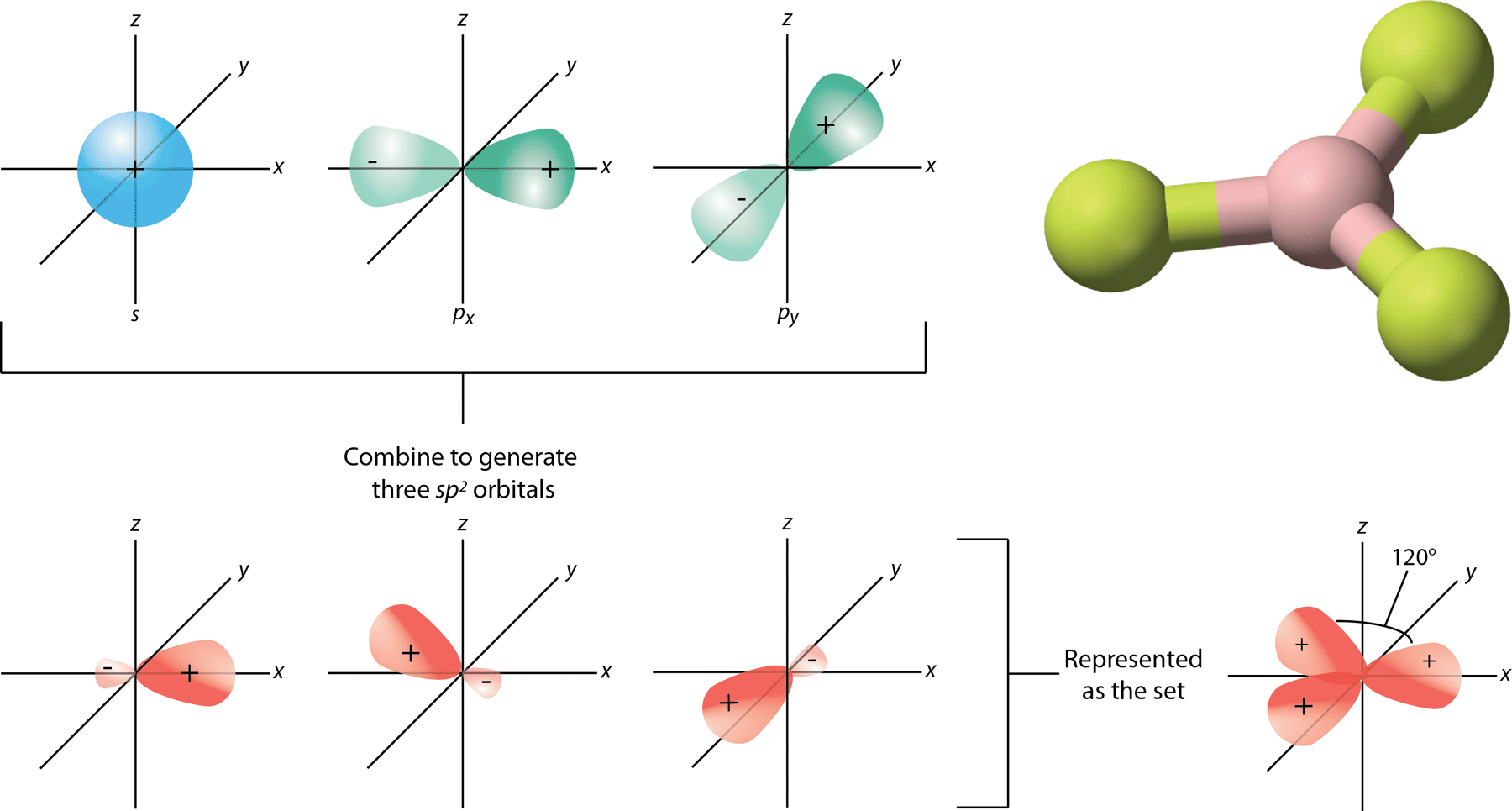
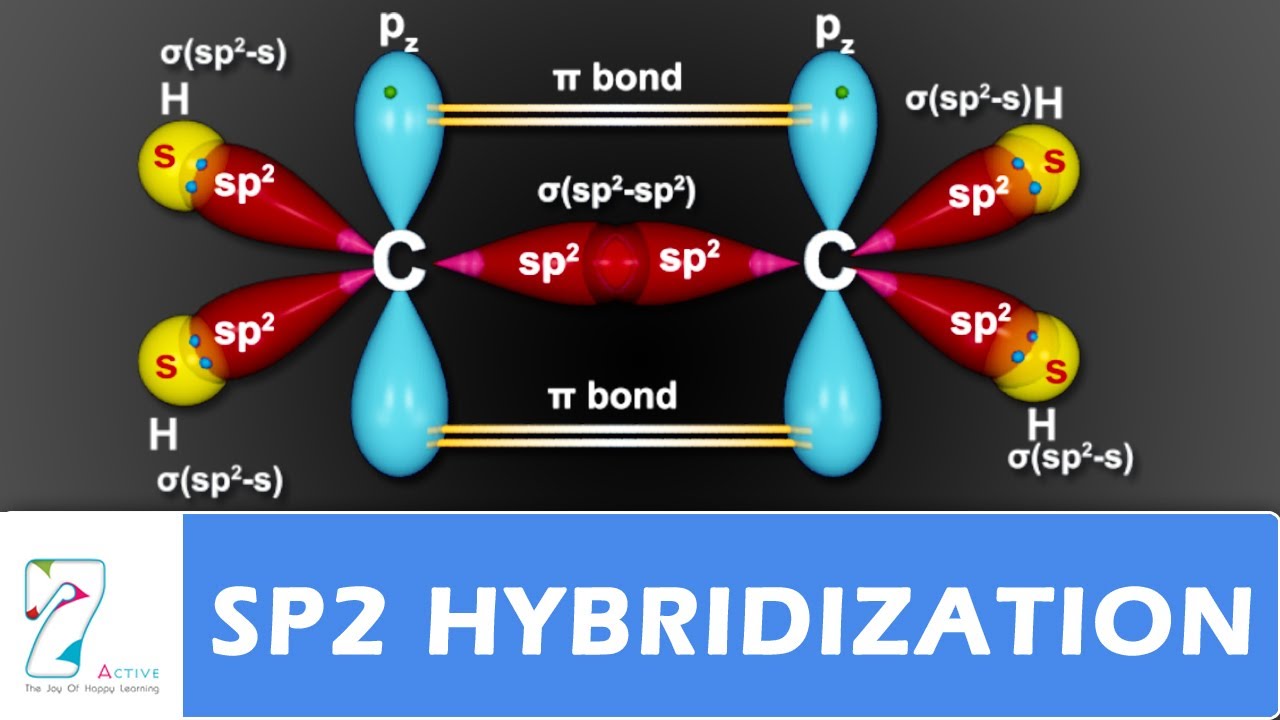
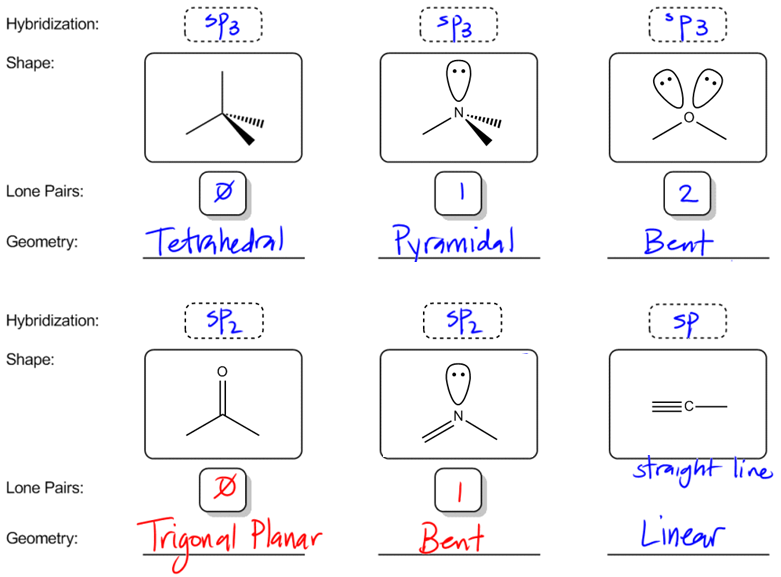
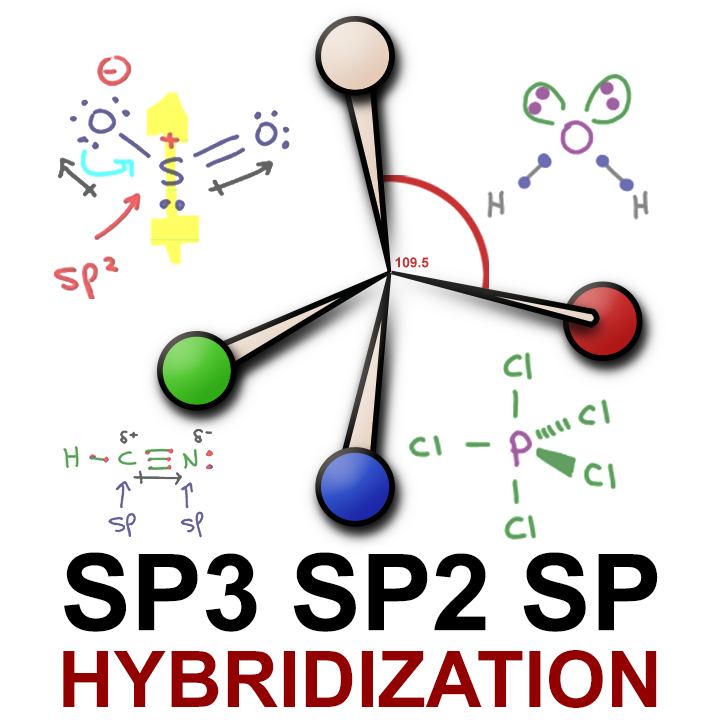
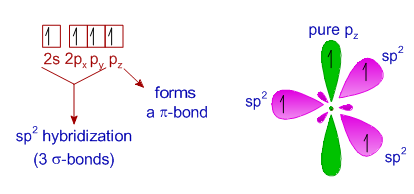
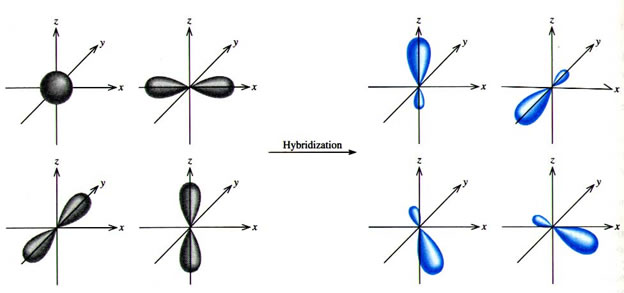
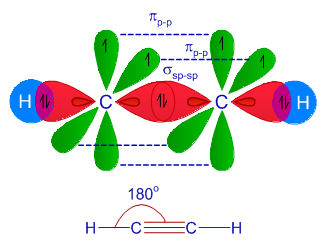
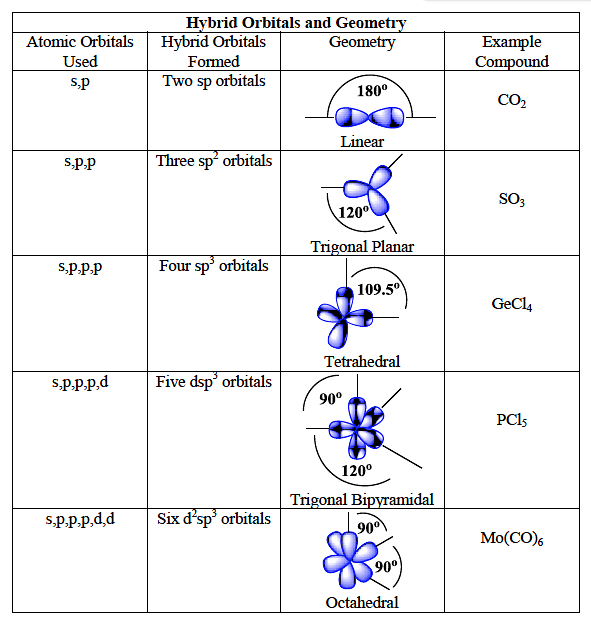
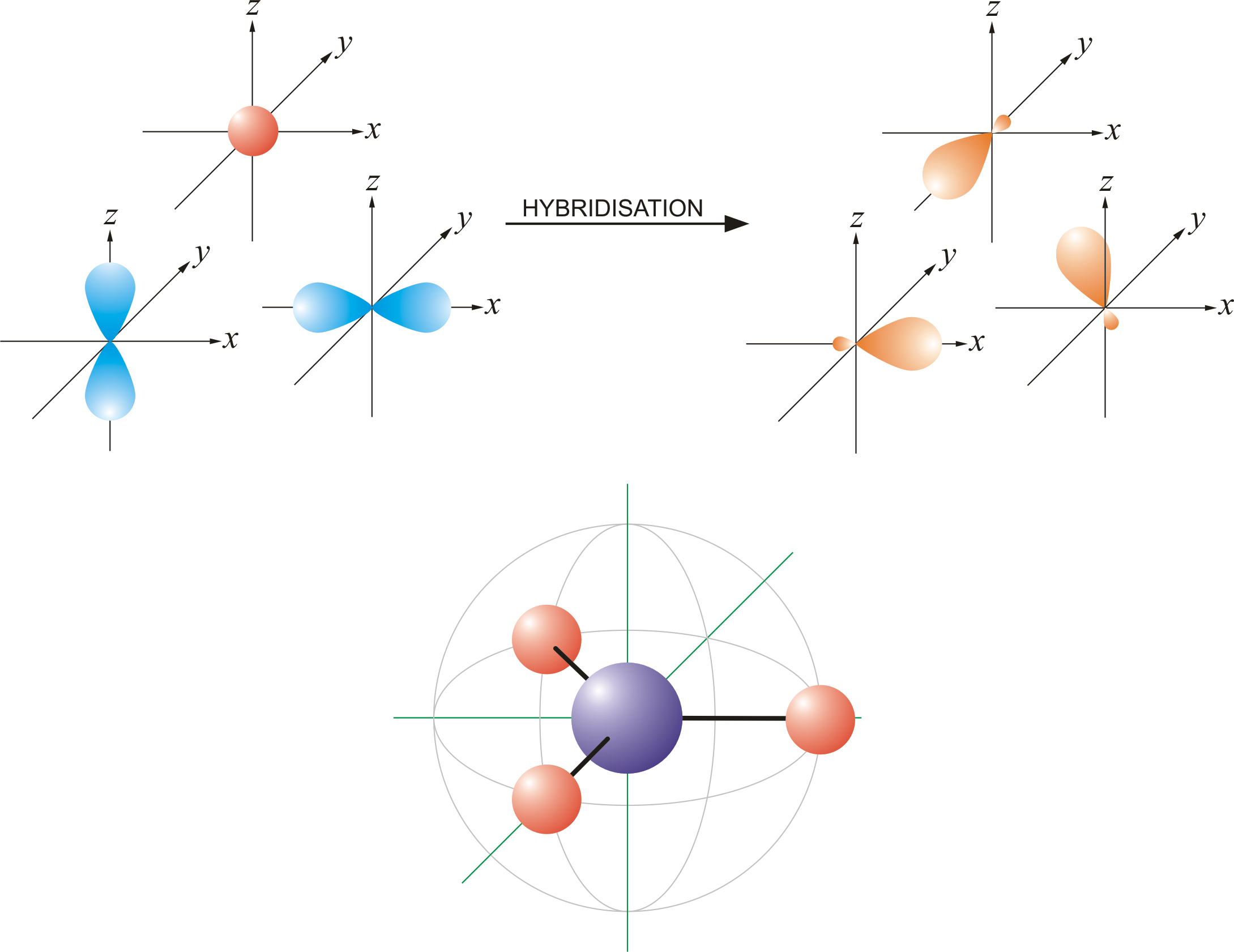
What are the shapes and bond angles of sp, sp2, sp3, sp3d, sp3d2 hybridised orbitals respectively? - Quora

Image result for hybridization sp sp2 sp3 and shape | Molecular shapes, Molecular geometry, Teaching chemistry
What are the shapes and bond angles of sp, sp2, sp3, sp3d, sp3d2 hybridised orbitals respectively? - Quora