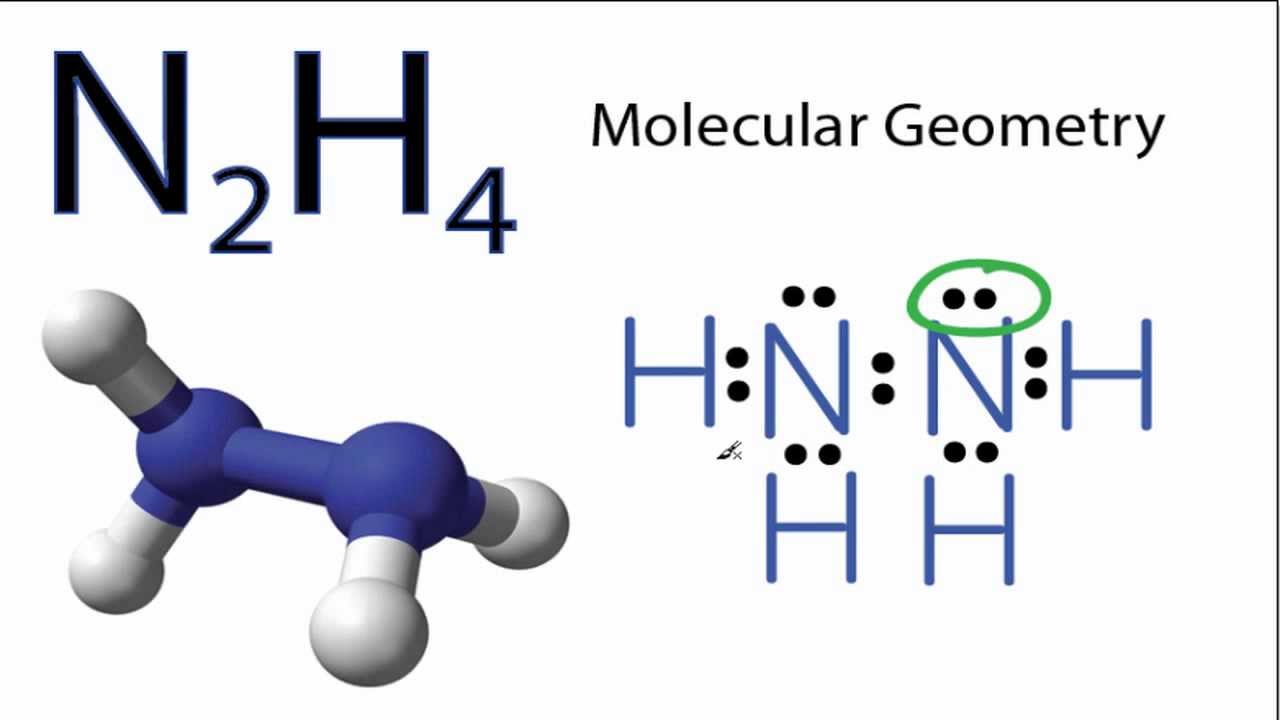
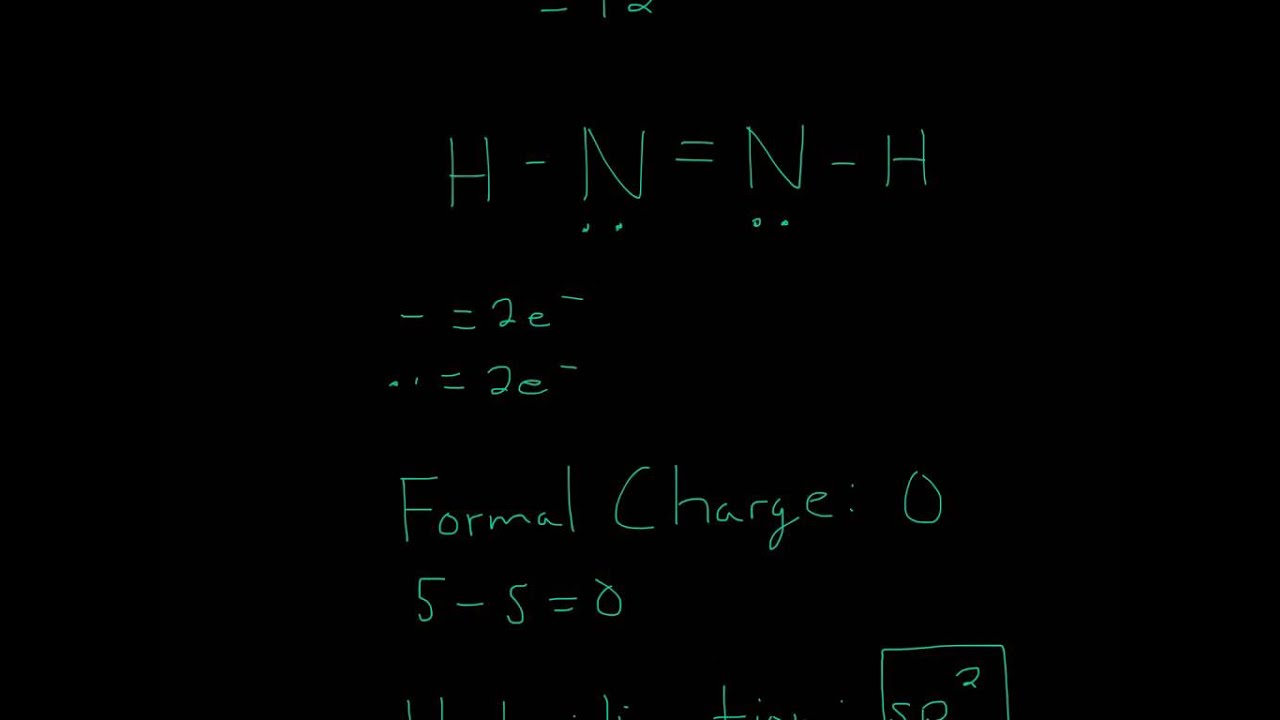Solved) - 9.62 The nitrogen atoms in N 2 participate in multiple bonding,... - (1 Answer) | Transtutors
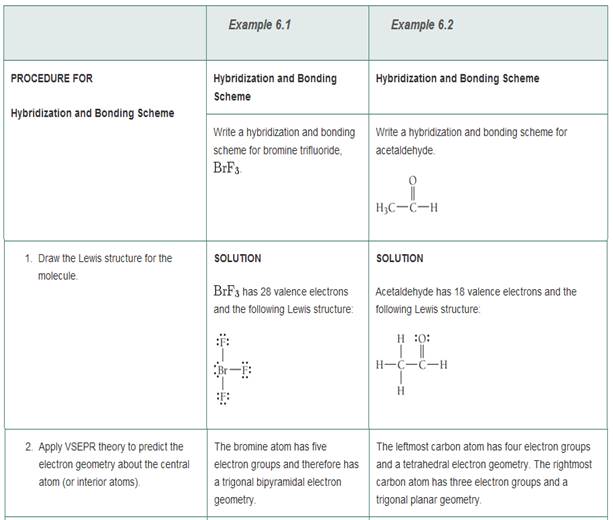
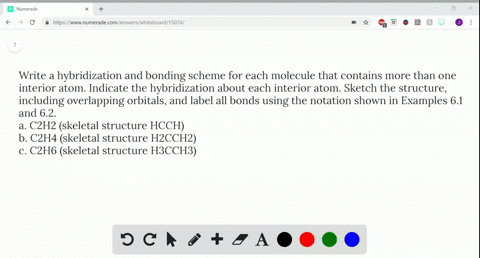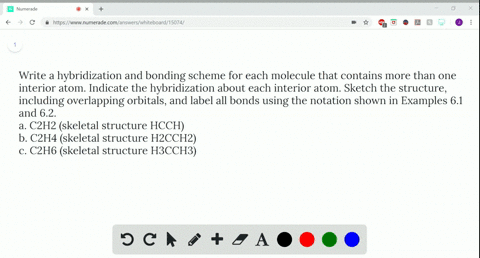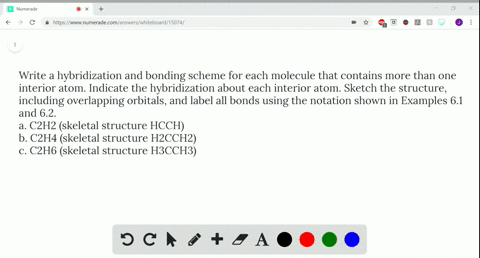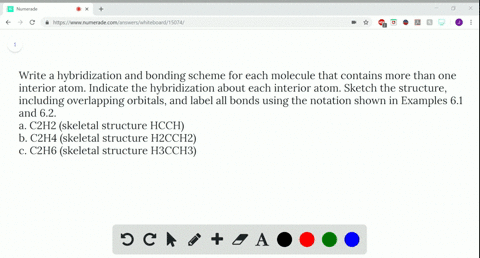
Write a hybridization and bonding scheme for each molecule that contains more than one interior atom. Indicate the hybridization about each interior atom. Sketch the structure, including overlapping orbitals, and label all

SOLVED: Write a hybridization and bonding scheme for each molecule that contains more than one interior atom. Indicate the hybridization about each interior atom. Sketch the structure, including overlapping orbitals, and label
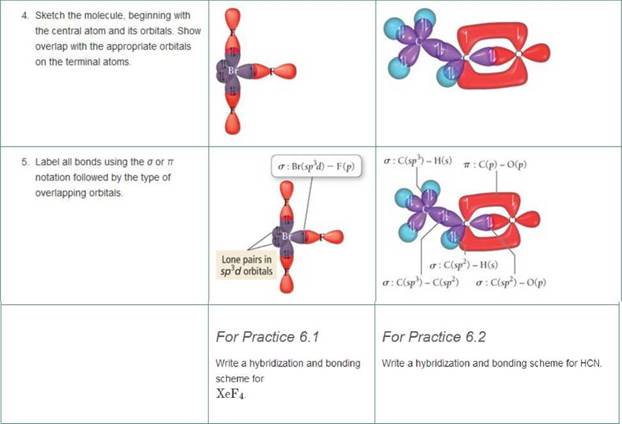
Write a hybridization and bonding scheme for each molecule that contains more than one interior atom. Indicate the hybridization about each interior atom. Sketch the structure, including overlapping orbitals, and label all

Karthik0000 assignment - 36 Determinetheelectrongeometry moleculargeometry andidealizedbondanglesforeachof thefollowingmolecules.Inwhichcasesdoyouexpect | Course Hero

Two structures can be drawn for cyanuric acid as follows.Give the hybridization of the carbon and nitrogen atoms in each structure. | Homework.Study.com
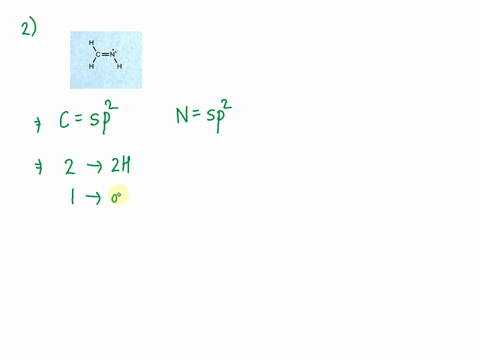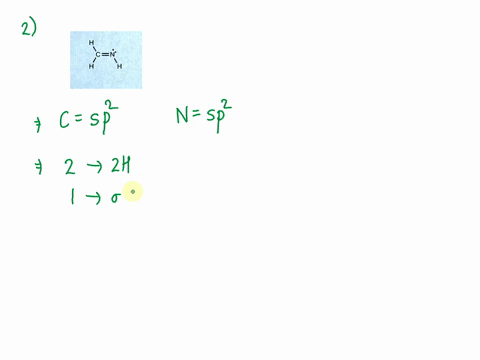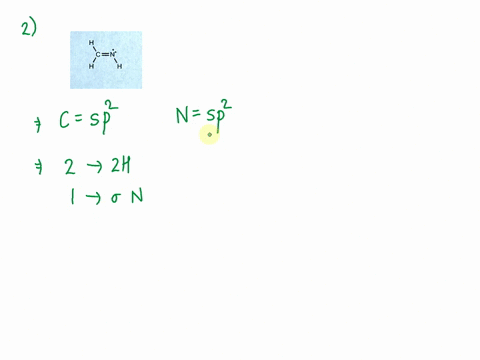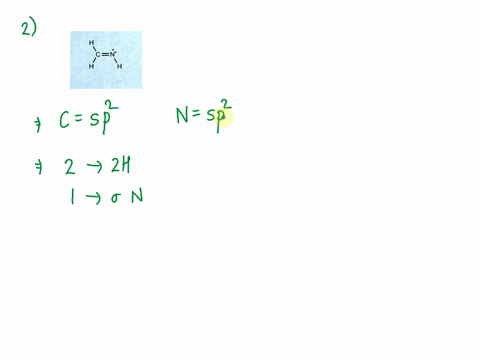
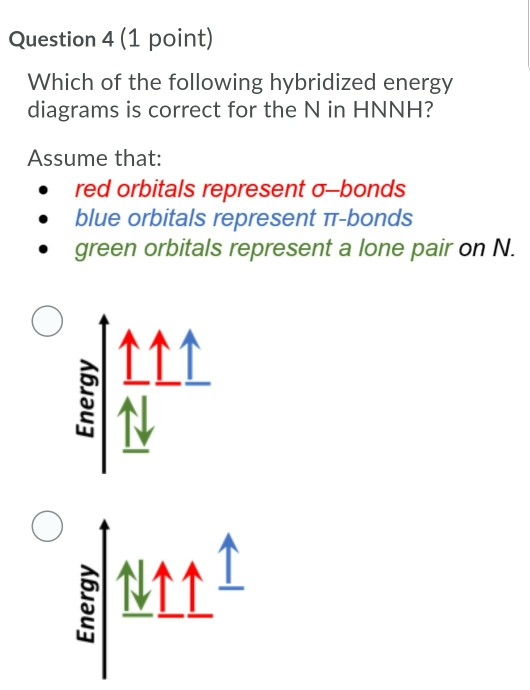
SOLVED: Consider the molecule with the connectivity H-N-N-H: The hybridization at each nitrogen atom is The double bond between the nitrogen atoms is made by the overlap of two hybridized orbitals, producing (

Experimental evidence suggests that the nitrogen atom in ammonia, NH3, has four identical orbitals in the shape of a pyramid or tetrahedron. Draw an energy-level diagram to show the formation of these

SOLVED: Consider the molecule with the connectivity H-N-N-H: The hybridization at each nitrogen atom is The double bond between the nitrogen atoms is made by the overlap of two hybridized orbitals, producing (
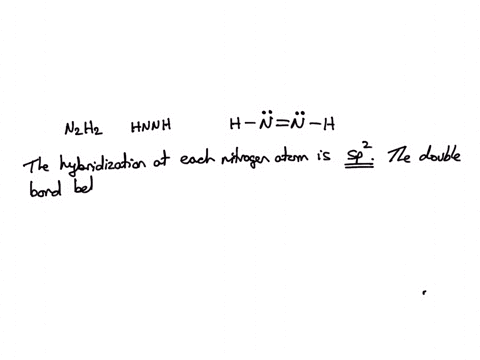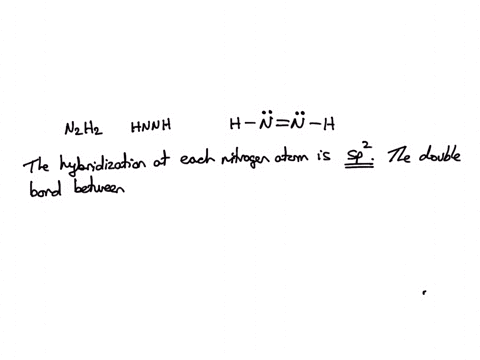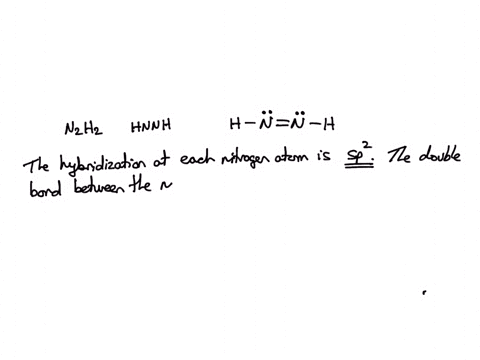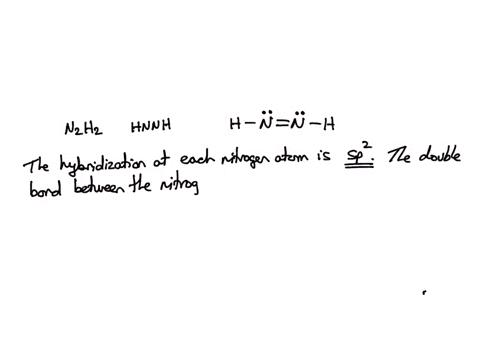



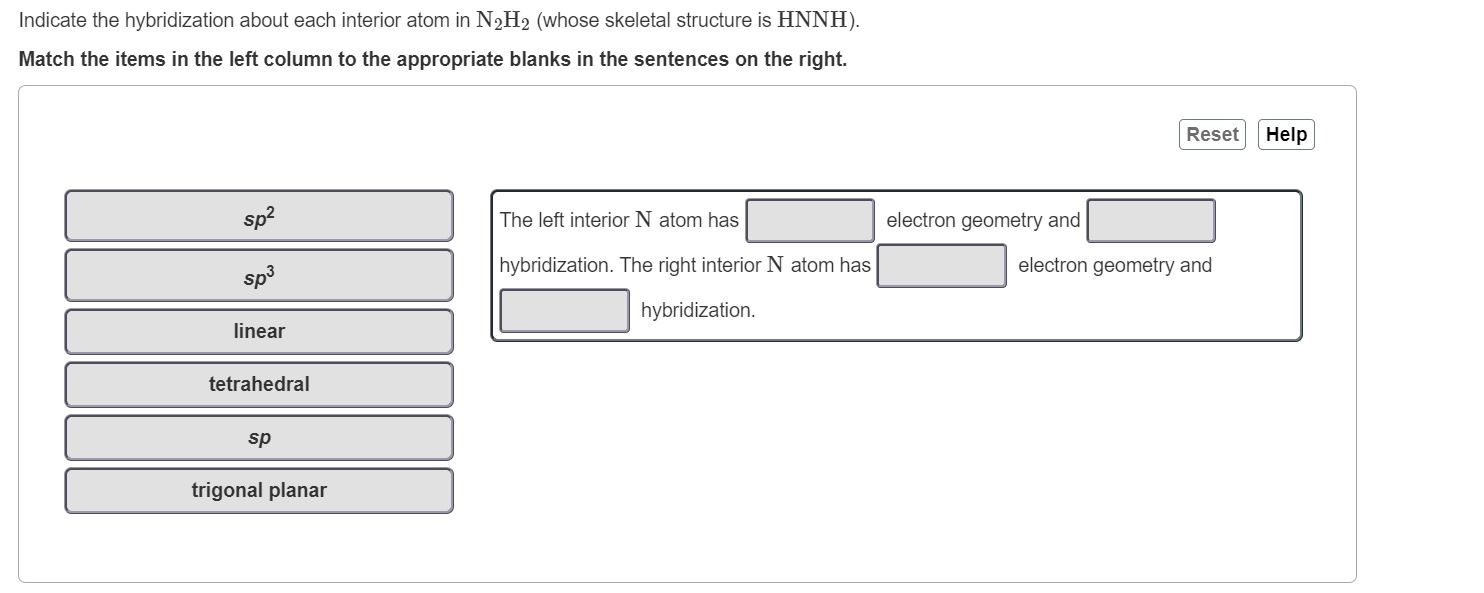
![ANSWERED] Part B Indicate the hybridization about each interior atom - Kunduz ANSWERED] Part B Indicate the hybridization about each interior atom - Kunduz](https://media.kunduz.com/media/sug-question-candidate/20220621022736275335-4634392.jpg)