
Draw an orbital picture of allene, H2C=C=CH2. What hybridization must the central carbon atom have to form two double bonds? What shape does allene have? | Homework.Study.com

8.26 | For the molecule allene, H2 C = C = CH2, give the hybridization of each carbon atom. Will the - YouTube

The hybridization of carbon atoms in C-C single bond of CH≡C-CH=CH_2 is: (a) s p^3-s p^3 (b) s p^... - YouTube






/chapter2/pages1and2/page1and2_files/sp2hyb.png)

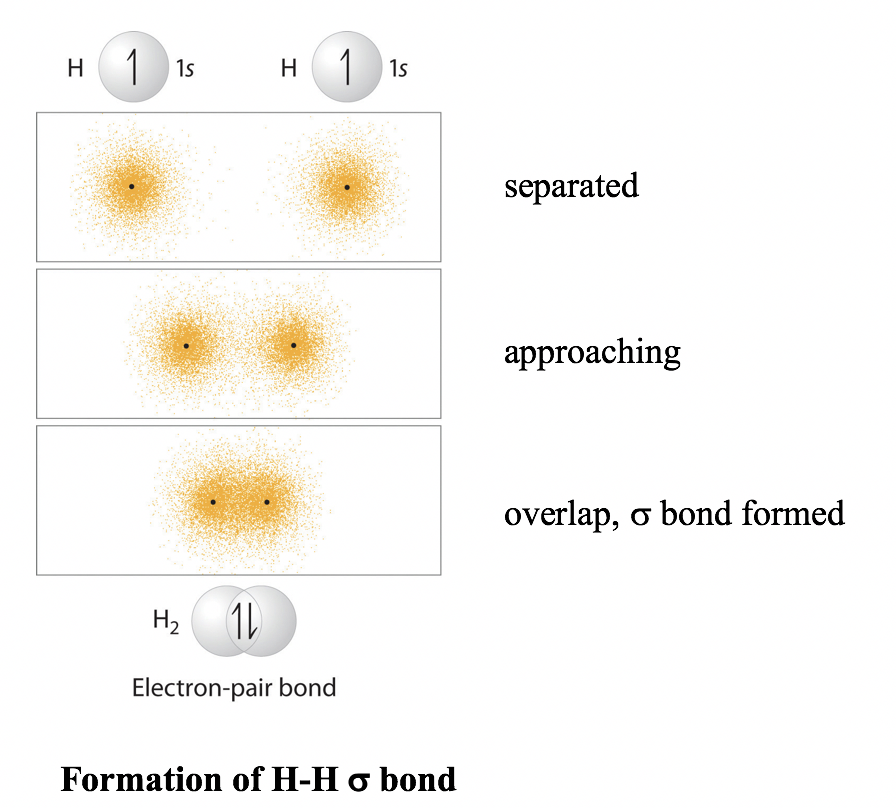


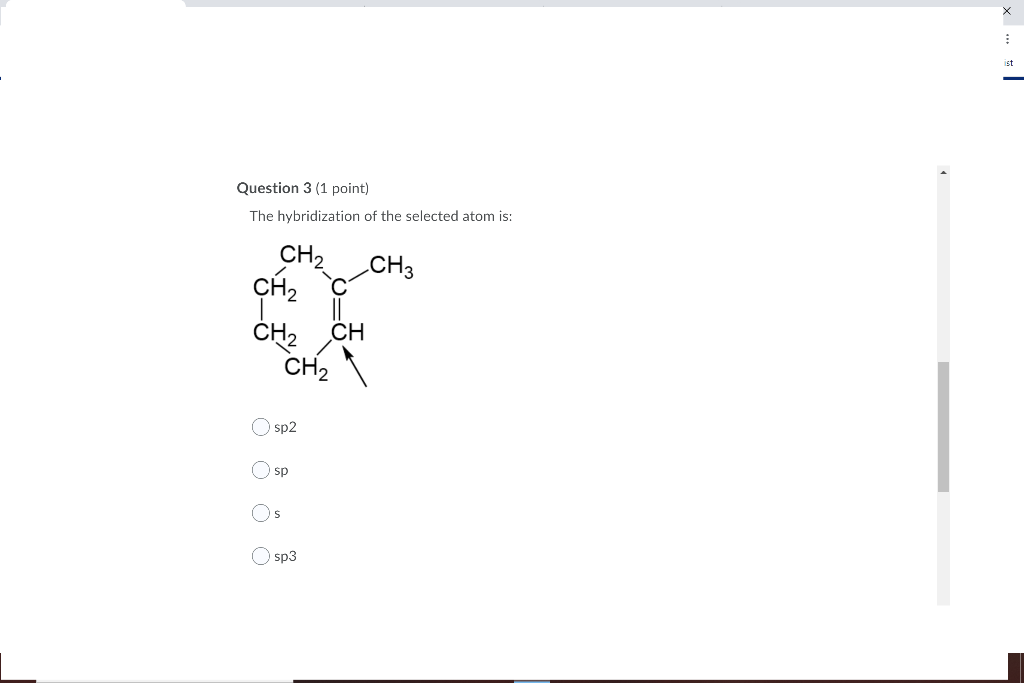
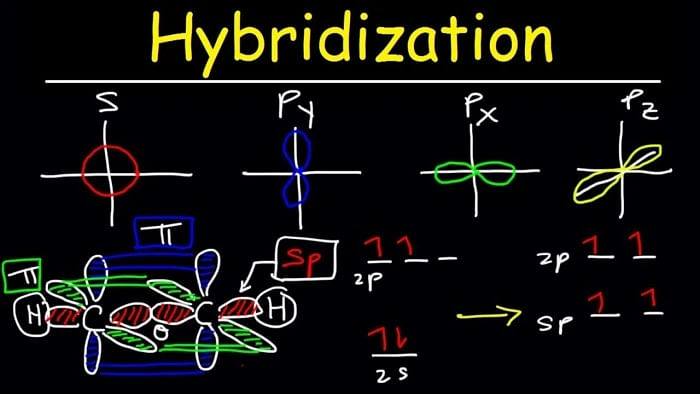

.jpg?revision=1&size=bestfit&width=601&height=192)